 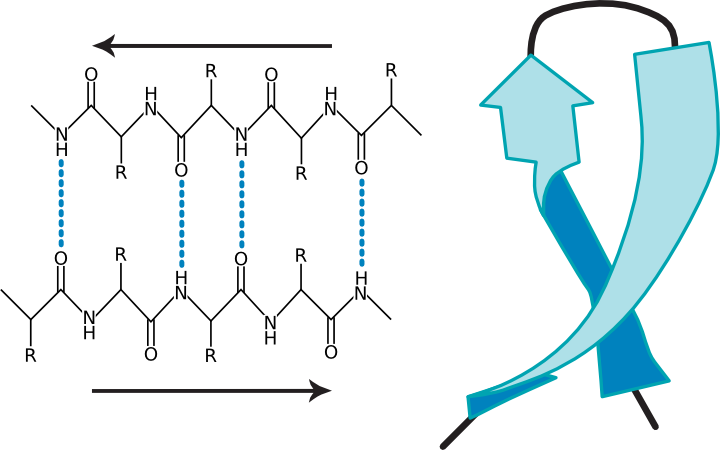
What remains highly enigmatic in the field of protein misfolding in general is the observation that peptides and proteins of no or of very diverse three-dimensional structure can form amyloids, in which the cross-β structure is the only common conformational motif 18, 19. Intermediates that are formed on the way include soluble oligomers, to which strong neurotoxicity is ascribed via different molecular mechanisms such as membrane disruption, receptor-binding followed by activation of signaling pathways or glutamatergic neuronal hyperactivation 7, 8, 9.ĭetails of Aβ fibril formation have been intensively investigated in the last two decades predominantly using solid state NMR and cryo-EM approaches in combination with molecular modeling, Structural models of the transient Aβ oligomers/protofibrils 10, 11, 12 and structures of Aβ fibrils 13, 14, 15, 16, 17 have been determined extensively. However, amyloid fibril formation does not follow only a single pathway, but occurs through several routes in the energy landscape 5, 6. The key molecular events underlying AD are fibril formation of amyloid β (Aβ) peptides and posttranslational modifications of the tau protein 3, 4. Progress in developing therapeutic treatment options is, among others, hampered by the long progression of the disease without any clinical symptoms, while moderate to severe levels of amyloid aggregates and neurofibrillary tangles in the brain result in the loss of neurons, disturbance of neuronal connectivity and in brain atrophy. In spite of intense research efforts invested into different aspects of this devastating form of dementia, AD is currently still incurable 2. The etiology of AD is complex and influenced by genetic, environmental, and/or lifestyle factors 1. The toxicological profile found for the variants depend on the type and extent of the modification.Īlzheimer’s disease (AD), being one of over 40 maladies caused by protein or peptide aggregation and misfolding, is a progressive and multifactorial disease leading to age-related changes in the brain. Using a multitude of biophysical methods, it is shown that these backbone modifications lead, in most of the cases, to alterations in the fibril formation kinetics, a higher local structural heterogeneity, and a somewhat modified fibril morphology without generally impairing the fibril formation capacity of the peptides. These mutations are expected to challenge the inter-β-strand side chain contacts as well as intermolecular backbone β-sheet hydrogen bridges. In our approach, a series of Aβ 40 peptide variants with two types of backbone modifications, namely incorporation of ( i) a methylene or an ethylene spacer group and ( ii) a N-methylation at the amide functional group, of the amino acids at positions 19 or 34 was applied. To this end, systematic modifications of the Phe 19–Leu 34 hydrophobic contact, which has been reported in almost all structural studies of Aβ 40 fibrils, helps understanding Aβ folding pathways and the underlying free energy landscape of the amyloid formation process. 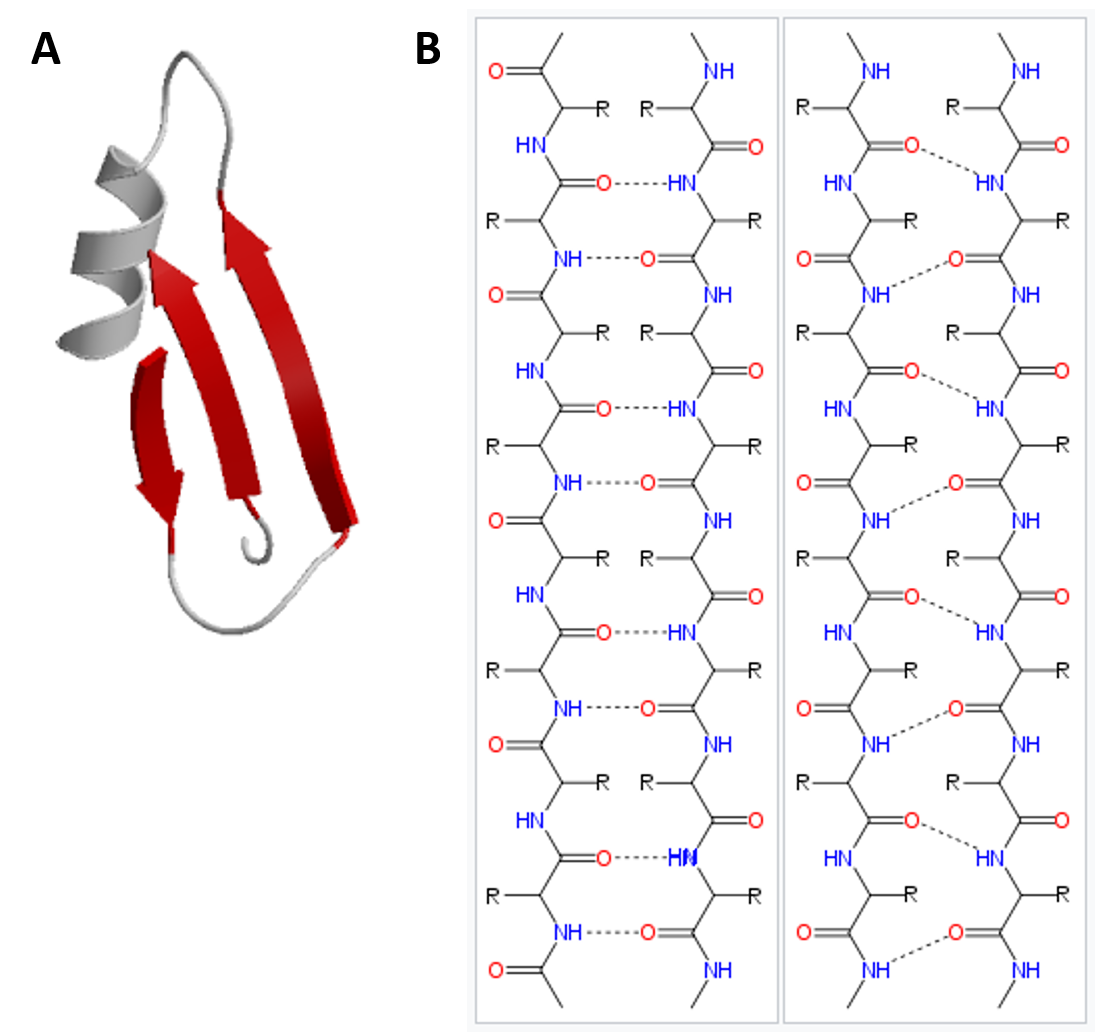
The pathway of formation and mechanism of action of Aβ aggregates in biological systems is still object of very active research. Fibril formation of amyloid β (Aβ) peptides is one of the key molecular events connected to Alzheimer’s disease. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
